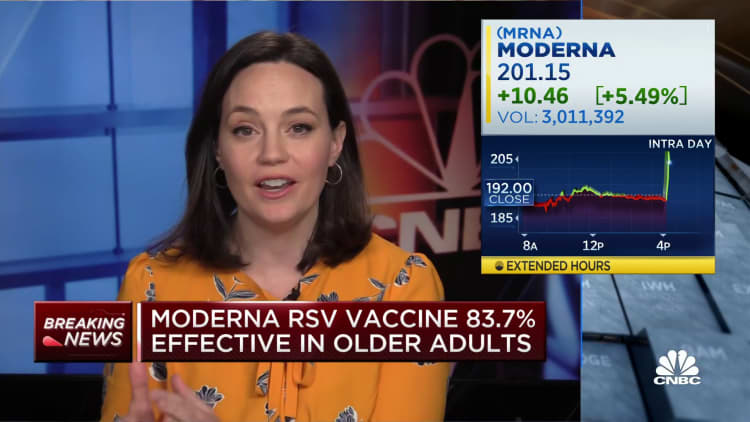
Moderna on Tuesday said its vaccine that targets respiratory syncytial virus is effective at stopping disease in older adults.
The vaccine was 83.7% effective at stopping lower respiratory tract disease, defined as two or more symptoms, in people ages 60 and older, in line with the Boston biotech company. It was 82.4% effective at stopping lower respiratory tract disease with three or more symptoms.
No safety concerns have been identified in the course of the clinical trial of the vaccine, in line with Moderna. The security and efficacy data from the trial shall be published in a peer-reviewed journal, in line with the corporate. The clinical trial has enrolled about 37,000 people across 22 countries.
Moderna said it plans to file an application for approval by the Food and Drug Administration in the primary half of this 12 months. There currently is not any FDA-approved vaccine for RSV.
Moderna’s stock rose nearly 7% in prolonged trading.

RSV infections kill between 6,000 and 10,000 older adults every 12 months and lead to 60,000 to 120,000 hospitalizations, in line with the Centers for Disease Control and Prevention.
The U.S. suffered an unusually severe RSV season in the autumn amongst children and older adults as the general public largely stopped practicing public health measures implemented in response to the Covid-19 pandemic, corresponding to masking and social distancing.
Moderna’s RSV vaccine uses the identical messenger RNA technology as the corporate’s successful Covid shots. The Covid vaccine turned Moderna into a worldwide name and delivered windfall profits, however it stays the corporate’s only commercially available product and demand is fading.
The Boston biotech company faces growing pressure to display that other products in its pipeline will successfully come to market. Morgan Stanley estimates the marketplace for an adult RSV vaccine is $7 billion to $10 billion.




