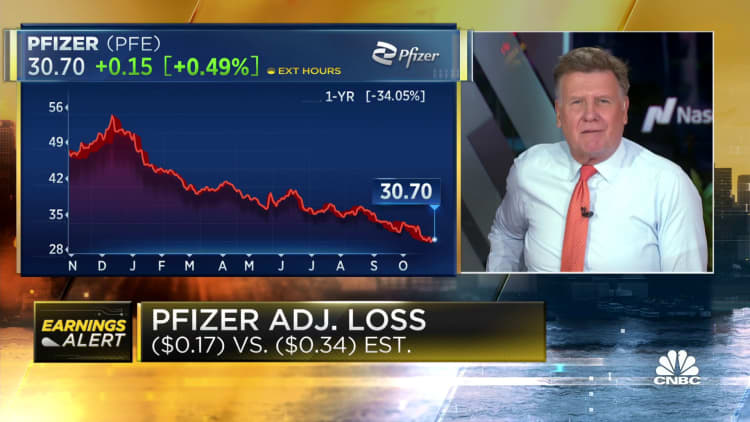
Pfizer on Tuesday reported a narrower-than-expected adjusted loss for the third quarter because the drugmaker recorded charges largely related to struggles for its Covid antiviral treatment Paxlovid and the Covid vaccine.
Pfizer said it recorded a $5.6 billion charge for inventory write-offs within the third quarter because of lower-than-expected use of Covid products. Of those previously announced write-offs, $4.7 billion is chalked as much as Paxlovid and $900 million is attributed to the corporate’s vaccine.
The pharmaceutical giant also reiterated the full-year adjusted earnings and revenue guidance it announced two weeks ago, which is drastically lower than its initial projections because of weakening demand for its Covid products. That decline in demand also led Pfizer to announce a sweeping $3.5 billion cost-cutting plan at the identical time.
Those efforts were seen as crucial to shore up investor sentiment as Pfizer and its rivals similar to Moderna struggle to navigate the rapid decline of their Covid businesses, that are transitioning to the industrial market within the U.S. this 12 months.
Here’s what Pfizer reported for the third quarter in comparison with what Wall Street was expecting, based on a survey of analysts by LSEG, formerly often known as Refinitiv:
- Loss per share: 17 cents, adjusted vs. 34 cents expected
- Revenue: $13.23 billion vs. $13.34 billion expected
Pfizer’s stock fell greater than 1% Tuesday. Shares of Pfizer were down roughly 40% for the 12 months through Monday’s close, putting the corporate’s market value at around $172.5 billion.
Pfizer reported third-quarter revenue of $13.23 billion, down 42% from the identical period a 12 months ago, because of the decline in sales of its Covid products.
The corporate’s Covid vaccine raked in $1.31 billion in sales, down 70% from the year-ago quarter. Analysts had expected the shot to usher in $1.53 billion in sales, in line with FactSet estimates.
Paxlovid posted $202 million in revenue, a drop of 97%. Analysts had expected $613.5 million in sales of the drug, in line with FactSet estimates.
Together, the products pulled in around $1.5 billion in revenue for the quarter. That compares with roughly $12 billion in sales in the course of the same period a 12 months ago.
For the third quarter, Pfizer booked a net lack of $2.38 billion, or 42 cents per share. That compares to a net income of $8.61 billion, or $1.51 per share, in the course of the same period a 12 months ago.
Excluding certain items, the corporate’s loss per share was 17 cents for the quarter. The inventory write-offs of Covid products accounted for an 84 cent per share adjusted loss, Pfizer CFO David Denton said during an earnings call on Tuesday.
Pfizer reiterated the guidance it outlined in October: The corporate expects 2023 sales of $58 billion to $61 billion and full-year adjusted earnings of $1.45 to $1.65 per share.
The corporate anticipates that its Covid vaccine will rake in $11.5 billion in sales this 12 months.
Meanwhile, the pharmaceutical giant expects its Covid antiviral treatment Paxlovid to usher in $1 billion in revenue. Pfizer has agreed to take eight million Paxlovid courses back early from the U.S. government, which is a component of an effort to get more higher-priced sales of the drug on the industrial market.
Denton said 2023 will inform the corporate’s expectations for vaccination rates and Covid product utilization rates within the U.S. next 12 months.
Pfizer’s non-Covid drugs
Excluding Covid products, Pfizer said revenue for the quarter grew 10% operationally.
The corporate said that growth was partly fueled by its latest vaccine against respiratory syncytial virus, which entered the market in the course of the quarter for seniors and expectant moms. The shot, often known as Abrysvo, posted $375 million in sales for the period.
Pfizer is “really pleased” with the performance of Abrysvo, which exceeded the corporate’s expectations, Chief Business Officer Angela Hwang said in the course of the earnings call.
She added that there’s “very fast uptake,” and Pfizer expects that momentum to proceed.
CFOTO | Future Publishing | Getty Images
Recently acquired drugs also drove revenue. Biohaven Pharmaceuticals’ migraine drug Nurtec ODT and Global Blood Therapeutics’ sickle cell disease treatment Oxbryta drew in $233 million and $85 million, respectively.
The corporate said revenue was also fueled by strong sales of Vyndaqel drugs, that are used to treat a certain kind of cardiomyopathy, a disease of the guts muscle. Those drugs booked $892 million in sales, up 48% from the third quarter of 2022.
A bunch of shots to guard against pneumococcal pneumonia also contributed, raking in $1.85 billion in sales for the quarter, up 15% from the year-ago period.
Meanwhile, Pfizer’s blood thinner Eliquis posted $1.49 billion in revenue for the third quarter, up just 2% from a 12 months ago. That got here in just below analysts’ estimates of $1.54 billion, in line with FactSet.
Eliquis, which is marketed in partnership with Bristol Myers Squibb, is among the many first 10 drugs to face Medicare drug price negotiations.
Wells Fargo analyst Mohit Bansal said in a research note Tuesday that the operational revenue growth in the course of the quarter “bodes well” for Pfizer to fulfill its full-year guidance of 6% to eight% growth for non-Covid products in comparison with 2022.
Pfizer drug pipeline, M&A
Pfizer is hoping to shift investor focus away from Covid toward its growth opportunities, including mergers and acquisitions and a record pipeline.
Pfizer CEO Albert Bourla noted that the corporate is nearing its goal of launching 19 latest products or drug indications in an 18-month span – a goal set last 12 months. Indications discuss with using a drug for a distinct disease type.
The corporate had a busy few months of product launches, which included a vaccine for RSV, an ulcerative colitis pill, a meningococcal vaccine and after all, the latest version of its Covid vaccine. That brings Pfizer to 13 out of 19 planned product launches.
Among the many six remaining product launches is Pfizer’s experimental flu vaccine, which it expects to launch after 2024. The corporate on Tuesday announced that its shot achieved positive initial results when put next to a currently marketed flu vaccine in an ongoing late-stage trial on people ages 18 to 64.
But investors are still waiting for more data on Pfizer’s flu vaccine in adults 65 and older. People 65 years and older are at higher risk of developing serious complications from flu, compared with young, healthy adults. Between 70% and 85% of seasonal flu-related deaths within the U.S. occurred amongst people 65 years and older in recent times, in line with the Centers for Disease Control and Prevention.
Investors are also waiting for updates on a midstage trial of Pfizer’s oral obesity pill danuglipron, which could potentially compete with Eli Lilly‘s experimental obesity pill orforglipron. Positive data could solidify Pfizer as a viable competitor in the load loss drug space, which Novo Nordisk and Eli Lilly have thus far dominated.
Pfizer executives said that the corporate expects to shut its $43 billion acquisition of cancer therapy maker Seagen in late 2023 or early 2024, subject to customary closing conditions similar to clearance by the Federal Trade Commission.
The European Commission, the chief body of the European Union, already approved the proposed buyout earlier this month.
Pfizer continues to consider the deal could contribute greater than $10 billion in risk-adjusted sales by 2030.
Don’t miss these CNBC PRO stories:




