David Ricks, CEO, Eli Lilly.
Scott Mlyn | CNBC
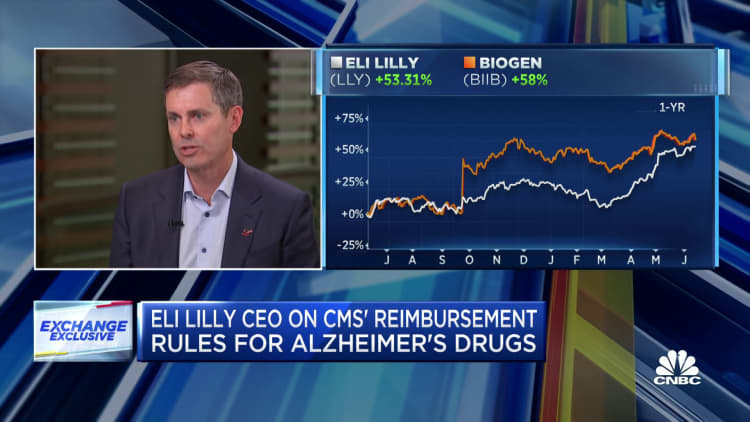
Eli Lilly CEO David Ricks on Tuesday said Medicare price negotiations, which aim to chop costs for older Americans, could potentially harm drug development.
“I’m really apprehensive in regards to the harm this may do to latest cures and possibilities in medicine,” Ricks said in an interview on CNBC’s “The Exchange.”
related investing news


Ricks was referring to a provision within the Biden administration’s Inflation Reduction Act that can allow the Medicare program to barter prices on the most expensive prescribed drugs annually.
He’s the newest pharmaceutical executive to publicly blast the availability and law at large, which is able to likely reduce company profits. Global drugmaker Merck last week sued the Biden administration over Medicare price negotiations in a bid to weaken this system.
Ricks said the “biggest problem” with the availability stems from a difference in timeline for negotiating prices on small-molecule drugs — meaning drugs fabricated from chemicals which have low molecular weight — versus biologic medicines, or those derived from living sources comparable to animals or humans.
Under the Inflation Reduction Act provision, Medicare can start negotiating prices on small-molecule drugs as early as nine years after they receive U.S. Food and Drug Administration approval, compared with 13 years for biologics.
Ricks said that distinction is “going to essentially truncate investment” in small-molecule drugs, that are “one of the vital efficient parts of health care.”
“We’ll get fewer of [small-molecule drugs] because investors are already saying to me, ‘Why would you put money into more small molecules when biologics get 13 years before negotiations?'” Ricks said.
Small-molecule drugs make up 90% of pharmaceutical drugs, in keeping with a study in ScienceDirect.
Novartis CEO Vas Narasimhan in February expressed similar concerns in regards to the various timeline, saying it is a top priority of the industry to correct the four-year gap between the 2 varieties of drugs, in keeping with Fierce Pharma.
One other provision of the Inflation Reduction Act requires pharmaceutical firms to refund Medicare through rebates if the costs of their drugs rise faster than the speed of inflation.
The primary set of eligible prescribed drugs was subject to Medicare inflation rebates starting April 1, in keeping with the U.S. Department of Health and Human Services.



