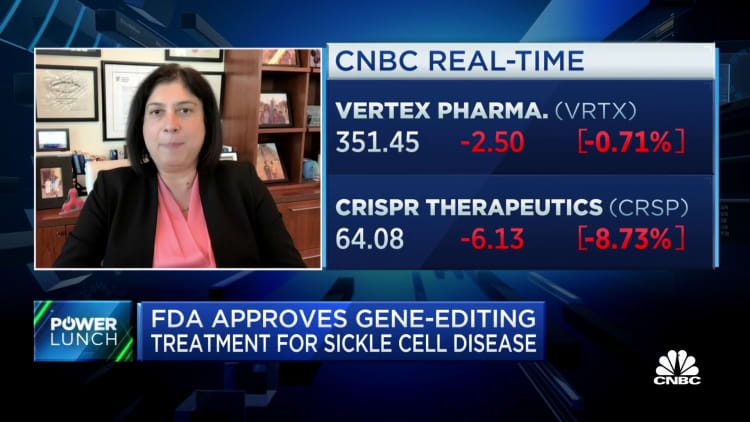
The U.S. Food and Drug Administration on Friday approved the country’s first gene-editing treatment, Casgevy, to be used in patients with sickle cell disease.
The approval comes a few decade after the invention of CRISPR technology for editing human DNA, representing a major scientific advancement. Yet reaching the tens of 1000’s of people that may benefit from the treatment could possibly be difficult given the potential hurdles — including cost, at $2.2 million per patient — of administering the complex therapy.
Casgevy, co-developed by Vertex Pharmaceuticals and CRISPR Therapeutics, uses Nobel Prize-winning technology CRISPR to edit an individual’s genes to treat disease. The treatment was approved by U.K. regulators last month.
Shares of Vertex fell 1% Friday, while shares of CRISPR fell 8%.
Sickle cell, an inherited blood disorder, causes red blood cells to develop into misshapen half moons that get stuck inside blood vessels, restricting blood flow and causing what are referred to as pain crises. About 100,000 Americans are estimated to have the disease.
This microscope photo provided on Oct. 25, 2023, by the Centers for Disease Control and Prevention shows crescent-shaped red blood cells from a sickle cell disease patient in 1972. Britain’s medicines regulator has authorized the world’s first gene therapy treatment for sickle cell disease, in a move that might offer relief to 1000’s of individuals with the crippling disease within the U.K.
Dr. F. Gilbert/CDC via AP, File
Casgevy uses CRISPR to make an edit to an individual’s DNA that activates fetal hemoglobin, a protein that normally shuts off shortly after birth, to assist red blood cells keep their healthy full-moon shape. In clinical trials, Casgevy eliminated pain crises in most patients.
The FDA approved the treatment for people 12 years and older.
“Sickle cell disease is a rare, debilitating and life-threatening blood disorder with significant unmet need, and we’re excited to advance the sphere especially for people whose lives have been severely disrupted by the disease,” said Dr. Nicole Verdun, director of the Office of Therapeutic Products throughout the FDA’s Center for Biologics Evaluation and Research, in a press release.
“Gene therapy holds the promise of delivering more targeted and effective treatments, especially for people with rare diseases where the present treatment options are limited,” Verdun added.
While the treatment itself is run just once, the entire process takes months. Blood stem cells are extracted and isolated before being sent to Vertex’s lab, where they’re genetically modified. Once ready, patients receive chemotherapy for just a few days to filter the old cells and make room for the brand new ones. After the brand new cells are infused, recipients spend weeks within the hospital recovering.
Vertex will take the lead on launching the drug and estimates about 16,000 individuals with severe cases of sickle cell will probably be eligible.
Even among the many individuals who may benefit probably the most, analysts worry few will clamor for a treatment that takes months to finish, carries the danger of infertility and could possibly be cost prohibitive. Vertex said in a regulatory filing Friday it would charge $2.2 million per patient for the treatment.
“We imagine the worth of drugs to reflect the worth that it brings, and the worth that this brings is a one-time therapy for potentially a lifetime of cure,” Vertex CEO Dr. Reshma Kewalramani said Friday in an interview with CNBC.
Vertex is seeing “unanimous enthusiasm” from payers, patients and physicians, because individuals with sickle cell have been marginalized, Kewalramani said, and the sphere hasn’t seen much innovation.
Since the procedure is so complex, it would be limited to certain health facilities like academic medical centers. Nine health-care facilities are ready to start out administering Casgevy, Vertex said in a release, with more facilities added in the approaching weeks.
Bluebird’s Lyfgenia
The FDA also on Friday approved a separate gene therapy by Bluebird Bio, called Lyfgenia that works otherwise than Casgevy but is run similarly and can also be intended to eliminate pain crises. That therapy was similarly approved for the treatment of sickle cell disease in people 12 years and older.
Bluebird will charge $3.1 million per patient for Lyfgenia. Shares of that company, which has a market value of nearly $300 million, fell 40% Friday.
Dr. Peter Marks, director of the FDA’s Center for Biologics Evaluation and Research, estimated during a call with reporters Friday that across the 2 therapies approved Friday, close to twenty,000 patients will probably be eligible for treatment.
However the FDA included a black-box warning – the strongest safety warning label – to Bluebird Bio’s Lyfgenia, noting that in rare cases the therapy may cause certain blood cancers.
The FDA added that warning after two patients who received Lyfgenia in a clinical trial died from a type of leukemia, Verdun told reporters Friday.
The agency said it’s still unclear whether Lyfgenia itself or one other a part of the treatment process, similar to the chemotherapy, caused the cancer.
But Marks said that the FDA wants patients to pay attention to all potential negative effects of your complete treatment process: “It’s in regards to the totality of the therapy that is given,” he told reporters.
Vertex didn’t see similar blood cancer cases in its clinical trial, which is why it didn’t receive a black-box warning on its label, Verdun noted.
Each Bluebird Bio and Vertex will follow patients who receive the treatments for 15 years as a part of a post-approval study. The FDA has encouraged the businesses to specifically monitor for malignancies, or the presence of cancerous cells that may spread to other sites of the body.




