Ozempic drug to treat diabetes.
Courtesy: Ozempic
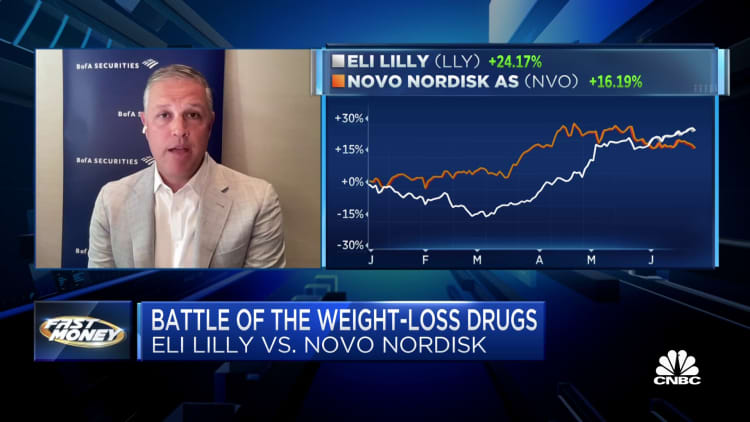
Of their most up-to-date earnings reports, Novo Nordisk and Eli Lilly proved why they’re the undisputed heavyweights within the prizefight for control of the rapidly growing weight-loss medications market. Beyond staggering sales figures for Novo’s Ozempic and Wegovy and Lilly’s Mounjaro, news of a study showing Wegovy can reduce the danger of heart disease and the anticipated approval of much more powerful pharmaceuticals to treat obesity will only strengthen the position of those venerable pharmaceutical giants, which have been in business for 100 years and 147 years, respectively.
Even so, their competitors usually are not ceding the market to the present leaders. Pfizer, Amgen and other pharmaceutical corporations are rushing to develop weight-loss drugs, though they might not be available for one more yr or more.
“We expect it’s Novo and Lilly, and everybody else is just scrambling to maintain up,” said Emily Field, head of European pharmaceutical equity research at Barclays, which predicts a $100 billion global marketplace for this class of medicine by around 2030. Since Wegovy and Mounjaro have been in the marketplace, “neither company could make the drug fast enough,” she said. “For each, demand has outpaced even their most bullish assumptions of what they’re in a position to supply.”
“It’s my top priority … expanding the capability of our ability to make not only Mounjaro but other drugs prefer it in our pipeline to satisfy the challenge here,” Lilly CEO David Ricks told CNBC’s Jim Cramer in August.
Ozempic and Wegovy are each GLP-1 agonists, substances originally formulated by Novo to treat type 2 diabetes. Besides controlling blood sugar, GLP-1s affect hunger signals to the brain, tricking the body into feeling full and slowing the speed at which the stomach empties. The sales growth has been so sharp that Novo Nordisk contributed nearly all of recent economic growth for its home nation of Denmark.
The Food and Drug Administration approved Ozempic in 2017 for diabetes and Wegovy in 2021 to treat obesity. Over time, each reduce body weight by about 15%. Mounjaro, introduced in 2022 to treat diabetes, incorporates GLP-1, plus GIP, an identical appetite suppressor that may result in weight reduction. All three drugs are prescribed as injectable pens that patients self-administer weekly.
On August 8, Lilly reported that its second-quarter income jumped 85% from the identical period a yr ago, driven largely by Mounjaro, which generated $979.7 million in sales for the quarter, in comparison with $16 million within the year-ago period and $569 million in the primary quarter of this yr. In December, analysts at SVB Securities projected that Mounjaro sales could reach $26.4 billion by 2030.
A month earlier, Lilly released data from a phase three trial of the drug, showing that it helped patients with obesity, though not diabetes, lose as much as 26.6% of their body weight after 84 weeks of treatment. Mounjaro is currently only approved by the Food and Drug Administration to treat diabetes, but the corporate has filed for FDA approval of Mounjaro specifically to treat obesity, which could come later this yr or in early 2024.
A pharmacist displays a box of Mounjaro, a tirzepatide injection drug used for treating type 2 diabetes made by Lilly at Rock Canyon Pharmacy in Provo, Utah, March 29, 2023.
George Frey | Reuters
Novo traded earnings jabs with its opponent on August 10, reporting that in the primary six months of this yr, sales of Wegovy soared 344% within the U.S. alone to just about $1.7 billion, while sales of Ozempic jumped 50% to greater than $3.7 billion. In response to financial analyst firm FactSet, sales of Wegovy and Novo’s other weight-loss injectable, Saxenda, could reach $6.1 billion before the yr is out and $15 billion annually by 2030. (Saxenda, in the marketplace since 2015, reduces weight by 6%-8%.)
Potentially larger news got here a few days earlier, when Novo released headline results of SELECT, a multi-year clinical trial of Wegovy, showing that it reduced the danger of major cardiovascular events similar to heart attacks or strokes by 20%, compared with a placebo. In the course of the earnings call with analysts, Novo CEO Lars Fruergaard Jorgensen said that the SELECT trial “underlines the importance of recognizing obesity as a serious chronic disease.”
The corporate expects to file for regulatory approvals of a label indication expansion for Wegovy within the U.S. and the European Union this yr, adding the drug’s cardiovascular advantages.
The search for brand new potential uses of those drugs with difficult health care issues continues, with the most recent headlines saying obesity drugs at the moment are being investigated as potential treatments for dementia and addiction, too.
Heart advantages, and doctor pressure, may force Medicare to cover
Results like these could prod government and personal insurers to reimburse more patients for the high price tag of weight-loss prescriptions. By law, Medicare hasn’t covered them since 2006, though some select Medigap and Medicare Advantage plans for retirees do. As of late July, Medicaid programs in nine states covered Wegovy, which costs $1,349 monthly. Private insurers often don’t cover GLP-1 drugs prescribed for weight reduction only.
Conversely, Medicare, Medicaid and most private insurers cover Ozempic ($936 per 30 days) when it’s prescribed for type 2 diabetes treatment, but not for weight reduction. Coverage for Mounjaro ($1,023 per 30 days) to treat diabetes varies based on a person’s insurance plan and drug advantages.
Based on the SELECT study — Novo will release full results later this yr — most payers may ultimately cover weight-loss drugs if a patient’s provider prescribes them to treat or prevent cardiovascular conditions, which may be exacerbated by diabetes and obesity.
“They’ll don’t have any selection at this point,” said Dr. Wajahat Mehal, director of the Yale School of Medicine’s Metabolic Health and Weight Loss Program, whose patients respond well to GLP-1s. The SELECT results “are going to place pressure on insurance firms” to cover those drugs, he said.
On July 20, a bipartisan group of U.S. senators and representatives reintroduced the Treat and Reduce Obesity Act, which might reverse the federal ban on Medicare coverage of obesity drugs. “With obesity rates on the rise in our country, we must do more to combat this epidemic head on,” said Democratic Senator Tom Carper of Delaware. “Too lots of those in need are being denied care due to high cost of medicines or inaccessible treatment options.” Added Republican Senator Bill Cassidy of Louisiana, “Expanding Medicare coverage to the treatments patients need enables them to enhance their health and advantages us all.”
Novo and Lilly have lobbied Congress on behalf of the laws, originally introduced in 2021, and have endorsed its reintroduction. Novo has also courted members of the Congressional Black Caucus and influential Black Americans to have Medicare cover weight-loss drugs. In response to the CDC, nearly 50% of non-Hispanic Black adults are classified as obese, the very best rate for any race or ethnicity.
The push by pharma giants for expanded coverage of those latest blockbusters comes because the federal government is for the primary time forcing drug corporations to negotiate with Medicare over a few of their most successful products, and a number of other recent news reports speculated that Ozempic will probably be a top goal for negotiated prices in the longer term. The pharmaceutical industry is difficult the brand new law’s constitutionality in court.
Attitudes about obesity being transformed
Overall, per the CDC, obesity within the U.S. affects 100 million (41.9%) adults and 14.7 million (19.7%) children and accounts for about $147 billion in annual health care costs. Historically, obesity has been considered a behavioral and lifestyle condition amongst people lacking the willpower to moderate eating habits and exercise recurrently. But those attitudes have been changing, not only in society at large but additionally amongst health care providers.
In 2013, the American Medical Association recognized obesity as a disease, which was an enormous step forward in the sector of medication, in addition to for pharmaceutical corporations to develop a latest generation of weight-loss drugs which might be simpler and safer than other obesity products, similar to Contrave, Orlistat, Qsymia and Imcivree.
“We have actually been in obesity research for greater than 25 years,” said Camilla Sylvest, executive vice chairman, business strategy and company affairs at Novo. “Plenty of things have modified in society within the meantime, not only that we’ve latest weight-loss products in the marketplace, but there’s also a much greater understanding of why obesity is a disease and why it must be treated.”
All that has increased the interest in treating obesity, “and due to this fact demand for Wegovy has exploded,” Sylvest said. Indeed, latest prescriptions for Wegovy and Ozempic have risen by 297% percent and 140%, respectively, since somewhat greater than a yr ago, in accordance with analysts at Cowen. What’s more, Novo and Lilly have each seen their stock prices greater than double prior to now three years.
It’s no surprise, then, that Novo and Lilly have had trouble maintaining with demand for his or her weight-loss drugs. In May, Novo reduced the U.S. supply of Wegovy starter doses to make sure access for existing patients, and in its recent earnings report said it expects that to proceed into the autumn. Likewise, in its second-quarter earnings call, Lilly executive vice chairman Mike Mason told analysts, “We do still expect to see tight supply and a few spot outages on Mounjaro through the tip of the yr.”
In 2021, Novo announced plans to speculate nearly $2.6 billion to construct three latest ingredients manufacturing facilities and to expand an existing production site in Kalundborg, Denmark. Then last May, the corporate committed $2.3 billion to expand its site in Hillerød, Denmark. In April, Lilly said it’s investing a further $1.6 billion in its two latest factories in Boone County, Indiana, bringing the corporate’s total commitment to the positioning to $3.7 billion.
Meanwhile, neither Big Pharma giant is resting on its weighty laurels, as each have increased their R&D budgets toward oral versions of their diabetes and obesity drugs, targeting patients who would like a pill as an alternative of a jab. Plus, pills taken once every day could also be cheaper to fabricate than single-dose pens used weekly.
Novo is in phase three clinical trials of oral versions of GLP-1, which it says may very well be even simpler in treating type 2 diabetes and obesity. It expects to file for FDA approval later this yr.
At an American Diabetes Association event in June, Lilly released phase two data for orforglipron, its first oral drug for obesity, saying it achieved as much as 14.7% weight reduction after 36 weeks. The corporate also released phase two data from trials on retatrutide, an injectable obesity medication that achieved as much as 17.5% weight reduction after 24 weeks.
Pfizer was testing two different oral drugs to treat type 2 diabetes and obesity, but is now specializing in one, danuglipron, after phase two trial results. “While we’ve our data from the kind 2 diabetes study [of danuglipron], we’re still studying the potential in obesity,” a Pfizer spokesperson said in an email. “When we’ve this information, which needs to be later this fall, we are going to make a plan for what the phase three [trial] will seem like.”
In December, Amgen reported that in phase one tests of its injectable obesity drug, AMG133, patients showed a weight reduction of 14.5% after 12 weeks of treatment. Phase two trials are ongoing, with data expected in 2024, and a product launch isn’t likely before 2026.
In order their competitors proceed to develop competitive therapies for diabetes and obesity, “Novo and Lilly will duke it out,” said Emmanuel Papadakis, an industry analyst at Deutsche Bank.
Once they will supply enough products to maintain up with the overwhelming demand, do not be surprised to see direct-to-consumer ads for Wegovy and Mounjaro popping up on the evening news. “Each corporations over time will probably be investing considerably to ascertain their marketing presence,” he said, “to the extent that it’s needed.”